Available imaging techniques cover a broad range from mass spectrometry (MALDI) to spectroscopy (FT-IR, TPEF, SHG, FLIM). Within the JBIL these different techniques are combined for multimodal imaging to help unravel major biomedical issues like the diagnosis of pathogens and host reply and molecular imaging of tumour tissue. Standard operating protocols for sample preparation for the different instruments are available upon request and will be provided by our staff after discussing your experimental requirements.
MALDI MS Imaging (MALDI MSI)
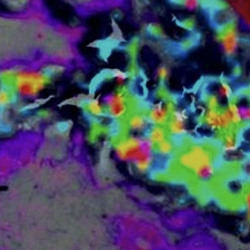 Matrix-assisted laser desorption/ionization (MALDI) mass spectrometry imaging (MSI), MALDI imaging in short, allows to obtain ‘omic’ profiles and identification of markers directly from thin frozen or formalin fixed paraffin embedded tissue sections. MALDI imaging is a label-free technique and can visualize the distribution of hundreds of molecular compounds (proteins, peptides, lipids) in a single, pixel wise measurement while maintaining the morphological and molecular integrity of the tissue and therefore allowing a subsequent histological staining of the same tissue section.
Matrix-assisted laser desorption/ionization (MALDI) mass spectrometry imaging (MSI), MALDI imaging in short, allows to obtain ‘omic’ profiles and identification of markers directly from thin frozen or formalin fixed paraffin embedded tissue sections. MALDI imaging is a label-free technique and can visualize the distribution of hundreds of molecular compounds (proteins, peptides, lipids) in a single, pixel wise measurement while maintaining the morphological and molecular integrity of the tissue and therefore allowing a subsequent histological staining of the same tissue section.
MALDI imaging is used for the pathological analysis of various tissue types, especially in oncology. A MALDI imaging data set includes several thousand individual mass spectra, with several thousand intensity values each. The superimposition of individual MALDI images with microscopic images or spectroscopic images can provide valuable and synergistic information.
The at the JBIL available instrument (UltrafleXtreme, Bruker) has a 2 kHz Laser for rapid measurement. Devices for paraffin removal, trypsin digestion and matrix application (Sun Digest and SunCollect, SunChrom) and software for evaluation and interpretation (Bruker and SCiLS Lab) are present.
Fluorescence Microscopy
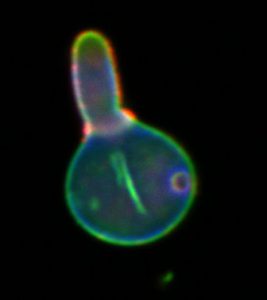
Fluorescence Microscopy is a versatile technique to investigate the distribution, interaction and dynamics of molecules and structures in cells and tissues. The molecule or structure of interest has to be labelled with a fluorophor to be detectable by fluorescence microscopy. This can be done in live cell imaging either by introducing target-specific label molecules or by transfecting plasmids encoding for autofluorescent fusion proteins. Using fixed biological material specific target structures can be visualised using immunofluorescence methods. With confocal laser scanning microscopy (CLSM) the maximal optical resolution in the imaging plane is about 300 nm and the axial resolution is about 800 nm. The available CLSM in the JBIL is housed within an S2 laboratory facility. Available excitation wavelengths include 405 nm, 458 nm, 488 nm, 514 nm, 561 nm, 633 nm. It also contains a Ti:Sa laser for two photon excitation (TPE) which is generally used to achieve a higher penetration depth in thicker biological samples.
Multispectral Optoacoustic Tomography
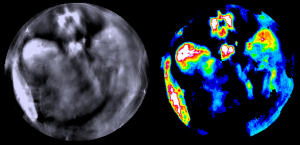
Multispectral Optoacoustic Tomography (MSOT) is a tomographic technique able to visualize molecules absorbing in the near and infrared spectra. Its main application is the quantitative differentiation of tissue by visualization of optical contrast and tissue biomarkers in vivo and in real time.
A tunable laser is employed to excite the specimens (which can also be small animals like zebrafish or mouse organs) at different wavelength. Especially (N)IR-dyes used for this technique as contrast agents, hemoglobin and oxygenated hemoglobin and water have very specific absorption bands in the IR-range, thus, their acoustic responses detected in MSOT serve to generate background contrast and gather anatomical b ut also functional information on tissue oxygenation. Fluorescent molecules will transform part of the energy into light, however the greater part of the energy will lead to a thermos-elastic expansion of the molecules, generating ultrasonic waves which are detected in MSOT by an array of ultrasound transducers. Since sound-waves are subjected to deep tissue scattering to a much lower extend than light this methods enables the generation of optical sections with a diameter of approximately 2.5 cm with a lateral resolution of 100 µm. The speed of the image acquisition allows the recording of videos at high frame rates to monitor e.g. heart rate. But also to study biodistribution, pharmacokinetics and elimination of (N)IR-absorbing molecules is feasible.
ut also functional information on tissue oxygenation. Fluorescent molecules will transform part of the energy into light, however the greater part of the energy will lead to a thermos-elastic expansion of the molecules, generating ultrasonic waves which are detected in MSOT by an array of ultrasound transducers. Since sound-waves are subjected to deep tissue scattering to a much lower extend than light this methods enables the generation of optical sections with a diameter of approximately 2.5 cm with a lateral resolution of 100 µm. The speed of the image acquisition allows the recording of videos at high frame rates to monitor e.g. heart rate. But also to study biodistribution, pharmacokinetics and elimination of (N)IR-absorbing molecules is feasible.
 The MSOT technology was realized by EFRE founds from the Thuringian State.
The MSOT technology was realized by EFRE founds from the Thuringian State.
S2 Laboratories
 Within the JBIL it is possible to work within S2 laboratories according to the Act on Genetic Engineering and the biological agents regulation. The S2 laboratories house certain microscopic systems like a CLSM with TPE and Raman microscopes.
Within the JBIL it is possible to work within S2 laboratories according to the Act on Genetic Engineering and the biological agents regulation. The S2 laboratories house certain microscopic systems like a CLSM with TPE and Raman microscopes.
Further Techniques
- Raman Spectroscopy including CARS, TERS
- Multimodal imaging, e.g. CARS, SHG, TPET